- Services
Our Work
 Discover how our language solutions have enabled Cama Group to sustain their international operations in the Uk, Germany, United States, China and Australia.
Discover how our language solutions have enabled Cama Group to sustain their international operations in the Uk, Germany, United States, China and Australia.
Our Work

We offer premium translation and localization services for manufacturers of medical devices that want to market their products across different countries.
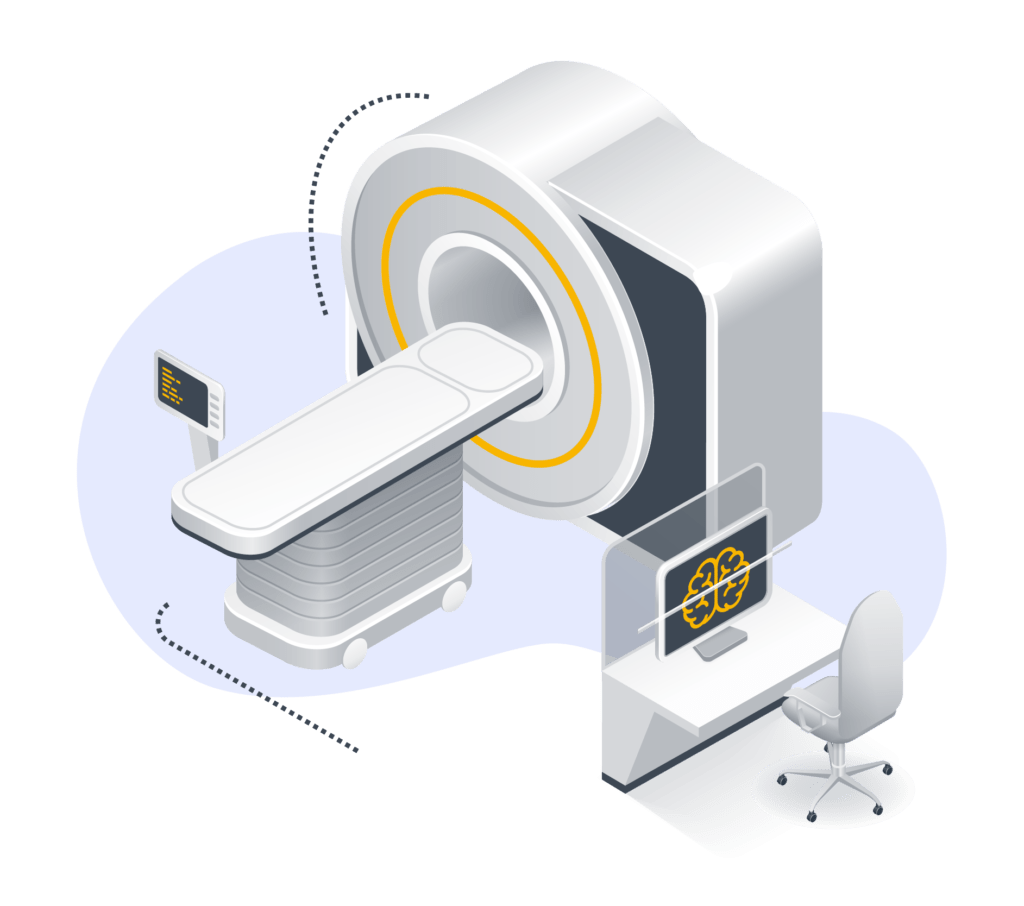
We’ve been supporting medical equipment manufacturers for over 15 years, always keeping up-to-date on the latest innovations and regulations related to the medical device sector in Europe and the rest of the world.

Wearable technology

Catheters

Patient monitoring

Diagnostic imaging systems
Dental equipment

Ophthalmic

Cardiovascular / Endovascular

Orthopedics

Respiratory
In the European Union, manufacturers can only place a CE mark on their devices once it has passed a conformity assessment. Since the introduction of EU MDR and IVDR, a medical device can only be approved for sale if all instructions and specifications meet strict language and translation criteria.

There are more than 500,000 different types of medical devices in the world. Whether it’s wheelchairs, glasses, pacemakers, electrocardiograms, or catheters, we’ve got you covered with our 360° language solutions.
We translate your content from one language into another (+140 languages). We work with any file format and have a curated network of linguists specialized in different fields.
Quality is essential in patent translations, that's why we only work with native translators that are specially chosen based on their experience and expertise in the field.
Through our desktop publishing services, we ensure the heavily formatted content such as packaging and labels meets the desired layout requirements for distribution.
When properly managed, corporate terminology can become a powerful asset. We help you create or maintain your existing multilingual terminology.
We help you deliver consistent onboarding & training programs to your teams across different geographies & in multiple languages.
When you need to localize highly creative content such as ads or videos, you can rely on our experts on transcreation, subtitling, and dubbing.
Today, most medical devices are equipped with embedded software that adds features, offers configuration options and makes them more accessible. Because the software user interface affects the interaction between the medical device and whoever is operating it, manufacturers also need to ensure that every string is accurately translated and localized.
Studies have found that over 80% of language translation quality issues are terminology-related, mainly inaccurate or inconsistent translations of medical terms in product package inserts, technical manuals (IFU), or user interfaces. Effective medical device terminology management in the source document saves businesses 20% or more on translation costs.

Via Napo Torriani, 29 – 20124 Milan Tel. +39 026 797951
Yellow Hub S.r.l. | Share capital: €50,000 | VAT: 00824050967 | Tax code: 06338220152